Endpoint PCR
IVD Polymerases
Diamond Taq® family polymerases show very good sensitivity and exceptional reproducibility. As highly thermostable enzymes, they offer an ideal solution for Molecular Diagnostic and demanding Research PCR & qPCR.
These Polymerases are purified from recombinant Escherichia bacteria containing the Thermus aquaticus DNA polymerase gene. This thermophilic eubacterium strain lacks TaqI restriction endonuclease and catalyzes 5’> 3’ synthesis of DNA with no detectable 3’> 5’ exonuclease activity. The enzyme has the 3’ extendase activity allowing TA cloning./p>
Ultra pure Diamond Taq® Benefits
Our IVD enzymes are highly suited for Diagnostic kits, Lab Services (with ASRs) and demanding Research
Exceptional Reproducibility
Stringent process and QC ensuring lot-to-lot & results reproducibility
Full
traceability
Quality Management System fully compliant to ISO 13 485 Medical Device standards and FDA’s Quality System Regulations
Customized fill
& finish
Cost-effective tailored solution
Ultra-low residual DNA content
<1 fg of genomic E. coli DNA / Taq Unit Typically < 0.01 fg or 0.002 E. coli genome copies /Taq Unit
Purity
Highly pure (> 98 %) and ultra-low bioburden (≤ 10 CFU/ml). Typically = 0 CFU/ml
Sensitivity
Amplification of DNA templates even at very low concentrations
How to select a polymerase?
| PCR | qPCR | Yield | Sensitivity Low Copy Template | Specificity | Difficult Template (GC & AT rich) | IP Burden | Free Sample | |
|---|---|---|---|---|---|---|---|---|
| Hot Diamond Taq® Short activation time |
+++ | ++ | +++ | +++ | +++ | +++ | + | TAQ-I032-100 |
| HGS Diamond Taq® Heat stability improved |
+++ | +++ | +++ | +++ | +++ | ++ | +++ | TAQ-I010-100 |
| Diamond Taq® | +++ | + | ++ | ++ | ++ | + | + | TAQ-I020-100 |
Hot Diamond Taq®
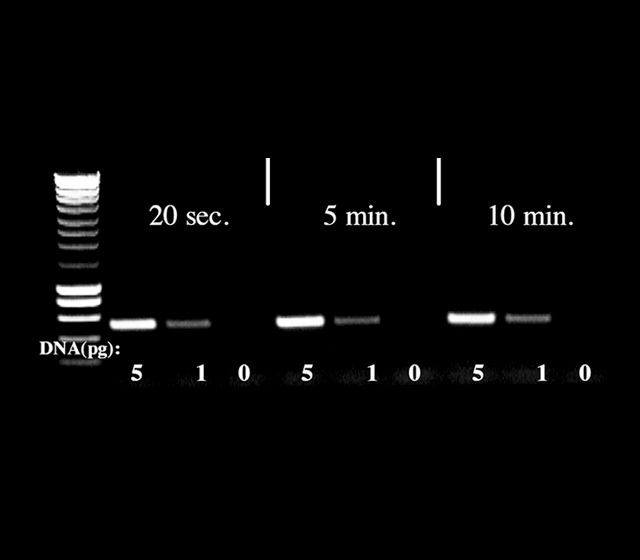
The Hot Diamond Taq® represents a completely new “HotStart Concept”. Neither accomplished through chemical modification nor a blocking antibody but a proprietary agent. This polymerase needs a really short activation time (20s are sufficient) and remains compatible with all existing protocols.
Fig: Hot Diamond Taq® was evaluated using from 20 seconds to 10 minutes as activation time for its ability to amplify 1 and 5 pg of λDNA. PCR products were analyzed by gel electrophoresis.
Traceability
All these enzymes, shipped on dry ice, follow the IVD traceability
at each step of the process, a Tracking number is sent to customer the day of the shipment.
Specifications
| Parameters | Specifications |
|---|---|
| Appearance* | Colorless solution |
| Identity (SDS-PAGE) | MW approx. 95 kDa |
| Volume activity | ≥ 5U/µl |
| Purity (SDS-PAGE) | > 98% |
| Performance test: PCR-λ DNA* | 0.5 kb fragment positive down to 5 pg |
| Performance test: PCR – 18S DNA* | 0.1 kb fragment positive down to 10 pg |
| Ribonucleases (up to 10U, 1h, 37°C) | Not detectable |
| Endonucleases, Exonucleases, Nicking activity (up to 30U, 16h, 65°C) | Not detectable |
| E.coli residual DNA | < 1 fg/Taq Unit |
| Bioburden* | ≤ 10 CFU/ml |
| Stability* | 24 months (at –20°C) from date of manufacture |
| Animal-derived additives* | None |
| HotStart | No detectable amplification without activation |
| Performance test PCR-Numb DNA (Hot Diamond Taq®) | 0.3 kb fragment positive down to 10 pg |