VHH Antibodies: Current and emerging applications in therapy, diagnostics, and drug delivery

This article reviews current and emerging applications of VHH antibodies and discusses key trends shaping their future clinical translation.
What are VHH antibodies?
Variable Heavy domain of Heavy chain (VHH) antibodies, also known as single-domain antibodies (sdAb), have rapidly evolved from niche research tools into one of the most promising antibody-derived formats in modern biotechnology. They are sometimes referred to as nanobodies® in the scientific literature, reflecting their small size and compact structure1.
Originally discovered in camelids such as llamas, camels and alpacas, VHHs correspond to the variable domain of heavy-chain-only antibodies, a naturally occurring class of immunoglobulins that lack light chains2 (Figure 1). This structural simplicity distinguishes VHHs from conventional monoclonal antibodies (IgGs), which are much larger (~150 kDa) and composed of multiple domains, and from antibody fragments such as single-chain variable fragments (scFv) that require paired variable regions to form a functional binding site.
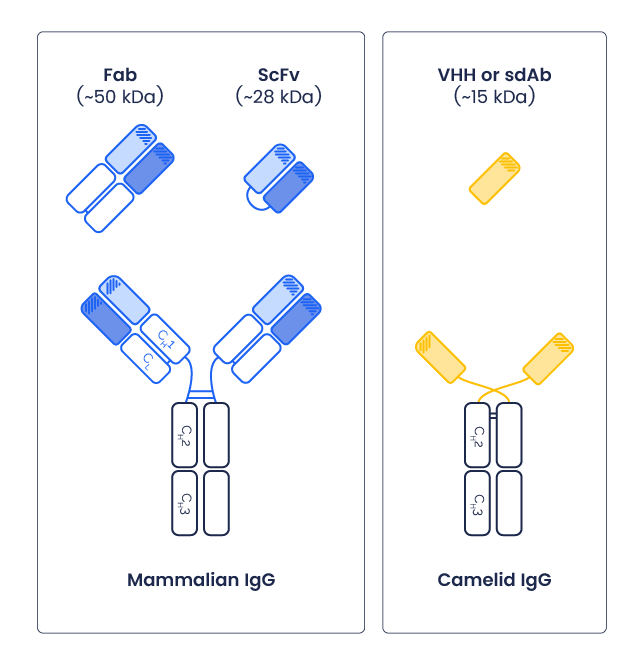
Figure 1: Comparison between mammalian IgG, Camelid IgG and their corresponding fragments.
Despite consisting of a single domain only (~12–15 kDa), VHH antibodies retain high target specificity and affinity while offering several distinctive biophysical advantages. Their small size and robust folding properties often translate into:
- enhanced stability under thermal or chemical stress
- high solubility and resistance to aggregation
- Ease of production in diverse expression systems
- improved access to cryptic or sterically restricted epitopes
- deeper tissue penetration, particularly in solid tumors or inflamed tissues, and, in some engineered formats, the ability to cross the blood–brain barrier (BBB) for central nervous system targeting.
These properties have been widely reviewed as compared to conventional antibodies3,4.
These characteristics make VHHs attractive not only as standalone therapeutic agents, but also as versatile engineering modules for next-generation biologics. In recent years, the field has expanded far beyond classical antibody replacement, with VHH domains increasingly integrated into multispecific constructs, fusion proteins, imaging probes, and targeted delivery systems5,6 (Figure 2).
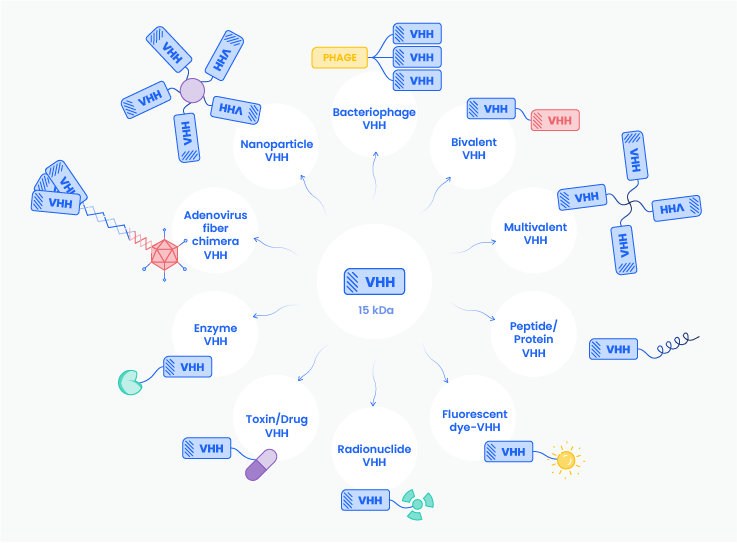
Figure 2: Illustration of VHH versatile usage. Inspired from Lezzi et al. 7
This article reviews the current and emerging applications of VHH antibodies in human and animal health, highlights key technological trends shaping their development, and discusses future directions as these molecules continue to move toward broader clinical adoption.
VHH antibodies in human and animal health
Clinical validation: Approved therapeutics and growing clinical pipeline
A major milestone for the VHH antibody field was the approval of Caplacizumab, the first nanobody-based therapeutic to reach the market. Caplacizumab targets the A1 domain of von Willebrand factor and is indicated for the treatment of acquired thrombotic thrombocytopenic purpura (aTTP), a rare and life-threatening thrombotic disorder characterized by microvascular platelet-rich clot formation. The clinical efficacy of caplacizumab was demonstrated in the pivotal HERCULES phase III trial8, and subsequent real-world studies have confirmed its therapeutic impact in routine clinical practice9.
Since this first approval, additional VHH-based therapeutics have been approved in different regions and disease areas. These include Envafolimab (anti–PD-L1) for advanced solid tumors in China10, Ozoralizumab (anti–TNF-α) for rheumatoid arthritis in Japan11 and Netakimab (anti–IL-17), an IgG-like antibody in which the VH domain is replaced by a VHH, approved for plaque psoriasis.
In parallel, VHH domains are also being incorporated into advanced therapeutic modalities such as CAR-T cell therapies, including Ciltacabtagene autoleucel for multiple myeloma, further expanding the clinical applications of single-domain antibody formats12.13
Therapeutic research in oncology, immunology, and infectious diseases
Beyond this first approval, the clinical pipeline of VHH-based therapeutics has expanded rapidly, with candidates being investigated in oncology, inflammatory disorders, and infectious diseases, reflecting growing confidence in the single-domain antibody format1,4.
Their compact size and high specificity enable efficient target engagement, particularly in contexts where tissue penetration and rapid binding kinetics are advantageous6.
In oncology, VHHs can function as direct tumor-targeting binders, recognizing tumor-associated antigens with high affinity. Their small molecular format may facilitate improved diffusion within solid tumors compared to conventional monoclonal antibodies, supporting their investigation in cancer immunotherapy strategies.
In infectious diseases, VHHs have also shown strong potential as neutralizing agents against viral pathogens, including rapidly evolving respiratory viruses, due to their stability and the ability to rapidly re-engineer binding domains when targets mutate 1,14.
While the clinical development of VHH antibodies has largely focused on human therapeutics, there is growing interest in their potential applications in animal health.
Recent work has demonstrated the feasibility of developing multivalent camelid-derived VHH constructs to neutralize veterinary-relevant toxins and pathogens. For instance, alpaca-derived VHH antibodies have shown strong neutralizing activity against tetanus neurotoxin, illustrating their potential as next-generation antitoxin strategies15.
Diagnostic and imaging perspectives
Beyond therapeutics, VHH antibodies have become valuable tools in diagnostics and molecular imaging. Due to their small size and high specificity, VHHs can serve as targeting vectors for imaging probes when conjugated to radionuclides or fluorescent labels.
In nuclear medicine, radiolabeled nanobodies are being explored for positron emission tomography (PET) and single-photon emission computed tomography (SPECT) imaging, particularly in oncology.
Compared to full-length monoclonal antibodies, VHH-based imaging agents often exhibit faster tumor penetration and more rapid clearance of unbound tracers from the bloodstream, enabling high-contrast imaging at earlier time points while reducing off-target exposure and contributing to favorable safety profiles16. Several radiolabeled nanobody-based tracers have demonstrated promising results in preclinical models and early-phase clinical imaging studies, supporting their growing role in precision cancer diagnostics17.
In addition to imaging, VHHs are increasingly integrated into diagnostic assays and biosensor platforms, benefiting from their stability and ease of recombinant engineering3.
Because VHHs can be produced recombinantly at scale and remain stable under variable storage conditions, they offer practical advantages for diagnostic deployment in agricultural and veterinary settings3.
In livestock and companion animals, infectious diseases remain a major cause of economic loss and welfare concerns, while also posing potential zoonotic risks. In this context, VHH antibodies are increasingly being explored as tools for rapid and robust veterinary diagnostics.
Emerging applications of VHH antibodies
Modular building blocks in multispecific and fusion biologics
Beyond their direct therapeutic applications, VHH domains are increasingly used as modular building blocks for engineered biologics. Unlike full-length monoclonal antibodies, VHH domains can be genetically linked together in a straightforward manner, enabling the design of bispecific and multispecific constructs with tailored functionality5.
In such formats, individual VHH domains can be combined to recognize multiple targets simultaneously. For example, one domain may bind a tumor-associated antigen, while another engages an immune cell receptor, thereby promoting immune cell recruitment and enhancing antitumor activity. This modular approach has become particularly relevant in cancer immunotherapy, where multi-target engagement is often required to achieve effective and selective responses 6.
In addition, VHHs are frequently fused to Fc domains or other protein scaffolds to extend serum half-life or introduce effector functions, illustrating the flexibility of single-domain antibodies as components of advanced engineered biologic formats1.

Targeted drug delivery systems
Beyond their use as therapeutic binders or multispecific modules, VHH antibodies are increasingly being explored as targeting vehicles in advanced drug delivery strategies. In these approaches, VHH domains guide therapeutic payloads toward specific cell types, improving selectivity and reducing off-target exposure4.
Because VHHs can be readily engineered and conjugated to other molecular entities, they have been incorporated into a variety of delivery platforms, including nanoparticles, liposomes, and antibody–drug conjugate-like constructs. Such systems aim to enhance the therapeutic index of cytotoxic agents or biologics by concentrating activity at the disease site rather than in healthy tissues.
In oncology, for instance, VHH-based targeting moieties are being investigated to deliver chemotherapeutic payloads more precisely to tumor-associated antigens, particularly through nanobody–drug conjugates and nanobody-decorated delivery systems, where VHHs guide payloads either directly or via targeted nanocarriers, supporting the development of more effective and better tolerated therapies1,18.
Innovative delivery strategies: mRNA and oral VHHs
In addition to classical injectable biologics, emerging research is exploring innovative delivery strategies that could further expand the versatility of VHH antibodies. Because of their compact structure and favorable biochemical properties, VHHs are being investigated in alternative therapeutic modalities beyond conventional antibody administration routes1.
Two particularly notable directions include:
- mRNA-encoded VHH therapeutics: In this approach, nucleic acid constructs are delivered to host cells, enabling transient in vivo expression of VHH antibodies. This strategy has been proposed as a rapid-response platform for infectious disease or immunotherapy applications, although it remains largely at the preclinical stage19.
- Oral or mucosal VHH prophylactics: Due to their high stability, VHH antibodies have been explored for oral or mucosal use in gastrointestinal infections, where they can bind and neutralize pathogens or toxins directly in the digestive tract. In such cases, their protective effect can occur locally, without the need for the antibody to enter the bloodstream 20.
Together, these early studies illustrate how the VHH format may support future therapeutic strategies that extend beyond traditional antibody delivery paradigms.
Key technological trends shaping VHH development
The VHH field is entering a phase of technological acceleration. Advances in design tools, delivery platforms, and scalable production systems are influencing how these molecules are engineered and translated into clinical applications.
AI-driven discovery and lead optimization
Advances in antibody discovery technologies are playing a central role in accelerating the development of VHH-based therapeutics. High-throughput sequencing, computational modeling, and machine learning approaches are increasingly used to identify and optimize VHH candidates more efficiently than traditional experimental screening alone1.
In particular, AI-guided methods can support the prediction of VHH structure–function relationships, helping researchers prioritize sequences with favorable binding properties as well as improved developability profiles. These approaches may reduce the number of iterative laboratory cycles required for affinity maturation and candidate selection21.
As computational antibody engineering continues to mature, it is expected to become an increasingly important component of early-stage VHH discovery and optimization pipelines. AI is also evolving beyond optimization toward de novo binder generation: recent workflows tailored for VHHs have produced epitope-focused binders that were experimentally validated, with structures consistent with predicted binding orientations, although many candidates still require affinity maturation to reach typical therapeutic affinity levels.13
Humanization and immunogenicity management
Because VHH antibodies originate from camelid immune systems, minimizing the risk of immunogenicity in humans remains an important consideration for clinical development. Although VHH domains share structural similarity with human antibody variable regions, their non-human sequence elements may still trigger anti-drug immune responses if not properly engineered3.
To address this, humanization strategies are commonly applied, involving the modification of framework residues to increase similarity to human germline antibody sequences while preserving antigen-binding affinity and stability. Such approaches have been widely discussed as essential steps in the translation of camelid-derived single-domain antibodies into safe therapeutic candidates4.
In parallel, advances in developability assessment and computational design are helping to integrate immunogenicity risk evaluation earlier in the VHH discovery process, supporting the selection of candidates with improved clinical suitability21.
Expression platforms and developability screening
As VHH antibodies progress toward clinical development, early attention to manufacturability and scalable production has become an increasingly important trend. One advantage of the VHH format is its compatibility with microbial expression systems, enabling scalable and cost-effective recombinant production compared to more complex antibody formats22.
Common platforms for VHH expression include Escherichia coli, which remains widely used for intracellular production, and the yeast Pichia pastoris (Komagataella phaffii), which enables secreted expression and can facilitate downstream purification while avoiding endotoxin-related concerns23,24.
In parallel, the increasing diversity of engineered VHH constructs has reinforced the importance of early developability screening, including evaluation of stability, aggregation propensity, and expression yield. Integrating these assessments early in discovery helps prioritize candidates most likely to succeed in scalable manufacturing and clinical translation21.
Conclusion: The growing need for reliable GMP manufacturing platforms
VHH antibodies have evolved from unconventional antibody fragments into a versatile class of biologics with expanding roles in therapeutics, diagnostics, and engineered delivery strategies. As clinical pipelines diversify and molecular formats become more complex, continued progress will depend not only on innovative design but also on robust developability assessment and scalable production pathways.
As more VHH candidates advance into toxicology studies and early clinical development, reliable GMP manufacturing platforms are essential to support the transition from discovery into clinical supply.
Eurogentec supports this translation by providing microbial GMP production of VHH drug substance in Escherichia coli or Pichia pastoris. Starting from a customer’s established research cell bank (RCB), Eurogentec’s dedicated platform enables delivery of GMP-grade VHH material within eight months, supported by an optimized toolbox approach and a defined quality control framework for clinical supply.
Key takeaways
- Multiple therapeutics incorporating VHH domains have now been approved globally, validating the clinical potential of this format across diverse indications and biologic architectures.
- VHHs are being developed across oncology, inflammation, infectious disease, and neurology, where their stability and tissue penetration offer advantages.
- Beyond therapeutics, VHHs are advancing in molecular imaging and diagnostic applications, including PET and SPECT tracers.
- Their modular structure enables the design of bispecific, multispecific, and fusion biologics.
- Emerging strategies such as mRNA-encoded VHHs and oral nanobody applications highlight the expanding versatility of the format.
- As pipelines mature, scalable and robust GMP production platforms are critical to translating VHH candidates into clinical-stage drug substance.
FAQ
What is a VHH antibody?
A VHH antibody is a single-domain antigen-binding fragment derived from camelid heavy-chain-only antibodies. Because it consists of only one variable domain (~12–15 kDa), it combines high specificity with robust biochemical stability3.
Are nanobodies used in approved drugs?
Yes. Several nanobody-based therapeutics have been approved globally, validating the clinical potential of single-domain antibody formats. These include caplacizumab8,9, the first approved nanobody drug, as well as more recent approvals such as envafolimab10 and ozoralizumab11.
Additional therapeutics incorporating VHH domains have also reached the market, such as netakimab13, an IgG-like antibody format, highlighting the broader use of VHHs across different biologic architectures.
VHH domains are also used in advanced therapeutic modalities, including CAR-T cell therapies such as ciltacabtagene autoleucel.12
Why are VHHs attractive for engineering multispecific antibodies?
Their compact and modular structure makes VHH domains easy to combine into bispecific or multispecific constructs, supporting advanced therapeutic designs, particularly in oncology and immune modulation5.
What expression systems are used for GMP VHH production?
VHH antibodies can be produced efficiently in microbial platforms such as E. coli and P. pastoris (K. phaffii), enabling scalable recombinant manufacturing for clinical and commercial supply 22.
What are emerging delivery strategies for VHH therapeutics?
Innovative approaches include mRNA-encoded VHH expression and oral or mucosal VHH prophylactics, leveraging the stability of single-domain antibodies for non-classical delivery routes1.
References
- Alexander, E. & Leong, K. W. Discovery of nanobodies: a comprehensive review of their applications and potential over the past five years. J Nanobiotechnol 22, 661 (2024).
- Hamers-Casterman, C. et al. Naturally occurring antibodies devoid of light chains. Nature 363, 446–448 (1993).
- Muyldermans, S. Nanobodies: Natural Single-Domain Antibodies. Annu. Rev. Biochem. 82, 775–797 (2013).
- Jovčevska, I. & Muyldermans, S. The Therapeutic Potential of Nanobodies. BioDrugs 34, 11–26 (2020).
- Amash, A. et al. Developability considerations for bispecific and multispecific antibodies. mAbs 16, 2394229 (2024).
- Cong, Y., Devoogdt, N., Lambin, P., Dubois, L. J. & Yaromina, A. Promising Diagnostic and Therapeutic Approaches Based on VHHs for Cancer Management. Cancers 16, 371 (2024).
- Iezzi, M. E., Policastro, L., Werbajh, S., Podhajcer, O. & Canziani, G. A. Single-Domain Antibodies and the Promise of Modular Targeting in Cancer Imaging and Treatment. Front. Immunol. 9, 273 (2018).
- Scully, M. et al. Caplacizumab Treatment for Acquired Thrombotic Thrombocytopenic Purpura. N Engl J Med 380, 335–346 (2019).
- Peyvandi, F. et al. Caplacizumab prevents refractoriness and mortality in acquired thrombotic thrombocytopenic purpura: integrated analysis. Blood Advances 5, 2137–2141 (2021).
- Markham, A. Envafolimab: First Approval. Drugs 82, 235–240 (2022).
- Tanaka, Y. Ozoralizumab: first Nanobody® therapeutic for rheumatoid arthritis. Expert Opinion on Biological Therapy 23, 579–587 (2023).
- Berdeja, J. G. et al. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study. The Lancet 398, 314–324 (2021).
- Evers, A., Guarnera, E., Pekar, L. & Zielonka, S. From discovery to the clinic: structural insights, engineering options, clinical, and ‘next wave’ applications of camelid-derived single-domain antibodies. mAbs 17, 2583210 (2025).
- Minatel, V. M., Prudencio, C. R., Barraviera, B. & Ferreira, R. S. Nanobodies: a promising approach to treatment of viral diseases. Front. Immunol. 14, 1303353 (2024).
- Sakamoto, C. et al. Complete Neutralization of Tetanus Neurotoxin by Alpaca-Derived Trivalent Nanobodies Aimed at Veterinary Medical Applications. Veterinary Sciences 13, 98 (2026).
- Xenaki, K. T., Oliveira, S. & Van Bergen En Henegouwen, P. M. P. Antibody or Antibody Fragments: Implications for Molecular Imaging and Targeted Therapy of Solid Tumors. Front. Immunol. 8, 1287 (2017).
- Yang, E. Y. & Shah, K. Nanobodies: Next Generation of Cancer Diagnostics and Therapeutics. Front. Oncol. 10, 1182 (2020).
- Ghosh, N., Sepay, N., Paul, M. & Im, J. Nanobody-Based Drug Delivery: Emerging Strategies for Targeted Cancer Therapy. IJN Volume 21, 1–47 (2026).
- Robinson, S. R. et al. An anti-Shiga toxin VHH nanobody multimer protects mice against fatal toxicosis when administered intramuscularly as repRNA. Infect Immun 92, e00239-24 (2024).
- Debatis, M. et al. Enteric pharmacokinetics of monomeric and multimeric camelid nanobody single-domain antibodies. PLoS ONE 18, e0291937 (2023).
- Ozdemir, E. S. et al. A Computationally Guided Approach to Improve Expression of VHH Binders. Biophysica 4, 573–585 (2024).
- Harmsen, M. M. & De Haard, H. J. Properties, production, and applications of camelid single-domain antibody fragments. Appl Microbiol Biotechnol 77, 13–22 (2007).
- De Groeve, M., Laukens, B. & Schotte, P. Optimizing expression of Nanobody® molecules in Pichia pastoris through co-expression of auxiliary proteins under methanol and methanol-free conditions. Microb Cell Fact 22, 135 (2023).
- Wu, Y. Trends in nanobody technology in industrialization. Discover Nano 20, 23 (2025).